Christian Brechot, MD, PhD
In the previous editions of our blog, we have emphasized the need to consider the microbiomes, not only humans but also animals, soils, and oceans, when discussing how our lives are being affected by our nutrition and our environment.
A new paradigm has emerged that might profoundly transform our approach to cancer and some chronic viral infections.
Indeed, microbiomes impact the outcome of infections by viruses and the response to anti-cancer therapies and their side effects! And this opens new avenues for improving the prevention and treatment of cancer and some dangerous chronic viral infections.
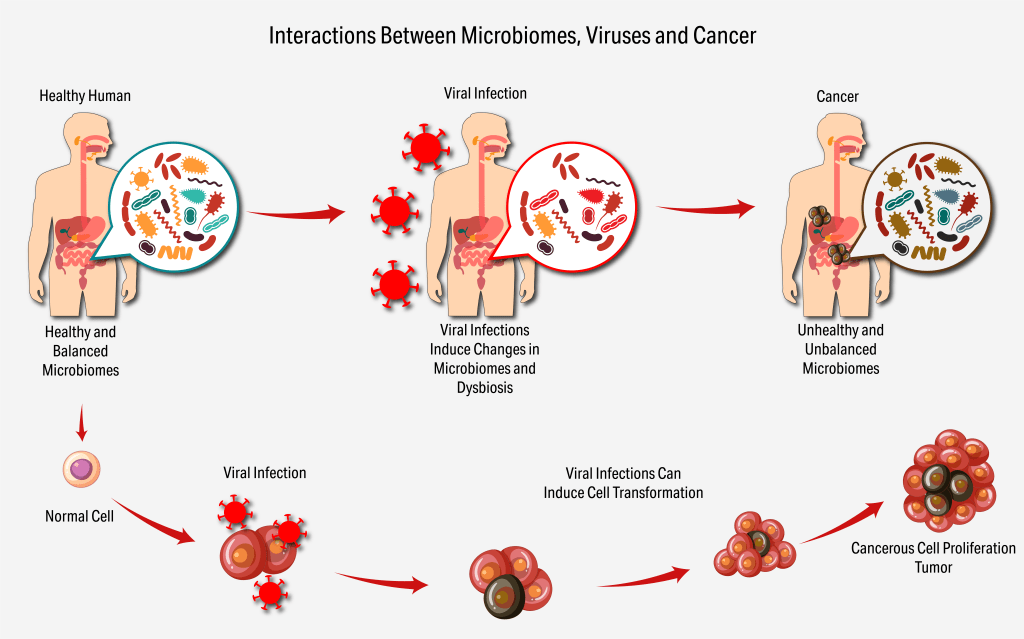
There is ample evidence that the gut microbiome shows unique microbial compositions among various cancers; also, it is clear that each tumor and its metastases exhibit an intrinsic microbiome that significantly impacts the tumor micro-environment and the overall progression of the cancer. Importantly, there is now evidence for the impact of such gut and tumor microbiomes on the response to immunotherapy and chemotherapy; as examples, transplantation of the intestinal microbiota from a patient who has responded to immunotherapy to a previously non responder patient can significantly improve his response to further therapy; this has been in particular shown for melanoma and lung cancer.
Treatment with immune checkpoint blockade (ICB) has markedly improved the survival of several cancers. However, a significant proportion of patients show primary or acquired resistance to treatment (up to 50% of patients with melanoma, and 25–44% of patients with non-small-cell lung cancer). In this context, the composition of the gut microbiome appears to be both predictive and prognostic of therapeutic response to ICB. These findings have led to microbiome-based treatment strategies to enhance clinical response to ICB and to mitigate their toxicity. Of note, some prebiotics-based strategies have been shown to improve the efficacy of some anticancer treatments. Regarding the intra-tumoral microbiome, advances in next-generation sequencing (NGS)techniques offer a more precise evaluation of the composition and function of the microbiome and its impact on the microenvironment. Yet, it is still unclear why certain intratumor bacteria promote tumorigenesis, while others trigger antitumor immune responses.

Moreover, there is now clear evidence that gut microbiome dysbiosis is an important factor driving the long-term complications of viral infections, such as, for example, HIV and HPV.
HIV
Many people living with HIV (PWH) are now achieving a life expectancy approaching that of the general population. This is related to the progress of effective antiretroviral therapies (ART). Yet, unfortunately, such patients frequently show accelerated aging with increased risk of age-related non-communicable diseases (NCD). Inflammation is the main driver of such complications and is, at least in part, related to gut dysbiosis. Indeed, HIV infection induces both disruption of gut-associated lymphoid tissue, microbial translocation, and an overall shift in gut microbiome composition, resulting in gut dysbiosis. HIV chronic infection is associated with the development of cancer. AIDS-defining cancers (ADCs: Kaposi’s sarcoma, non-Hodgkin lymphoma [NHL], invasive cervical cancer) are traditionally distinguished in HIV-infected patients; other cancers are referred to as non-AIDS-defining cancers (NADCs). NADCs, in turn, are usually classified into virus-related cancers (HPV-, EBV-, and HCV-related cancers) and virus-unrelated cancers
10–20% of all deaths of HIV positive individuals are attributable to cancer, and nowadays, people living with HIV still have a 1.6–1.7-fold greater overall risk of cancer development relative to the general population, and the risk is rising with age. This can be explained by the association of chronic inflammation, immunosuppression, and the frequent coinfection with other oncogenic viruses (Epstein-Barr Virus [EBV], Human Herpesvirus 8 [HHV-8], Human Papillomavirus [HPV], Hepatitis B and C Viruses [HBV, HCV).
Thus, understanding how gut dysbiosis, inflammation, and cancer development are connected is crucial for improving the health outcomes of PWH and identifying those at higher risk.
HPV
Nearly 90% of women are exposed to HPV; most infections resolve; however, about 10% persist, and this persistence is associated with increased risk of cervical cancer. Indeed, High-risk types like HPV 16 and 18 are the leading cause of cervical cancer. Importantly, HPV 16 and, to a lesser extent, 18 are associated with other cancers in men and women, such as head and neck (mostly oropharyngeal), anal and penis cancers
Several studies have now demonstrated that HPV infection is associated to dysbiosis of the vaginal and cervix, characterized by a low proportion of Lactobacillus and increased anaerobic bacteria. A dysbiotic cervicovaginal microbiome is more permissive to persistent HPV infection; it increases viral oncogene expression and integration of its genome, key steps in cervical carcinogenesis, and this eventually leads to cervical dysplasia and cancer. Dysbiosis fosters a pro-inflammatory environment, and chronic inflammation further exacerbates epithelial damage, thus favoring oncogenesis. Finally, dysbiosis also impairs mucosal barrier function, stimulating local inflammation and further promoting viral persistence and genome integration.
Of note, the Microbiota of Hispanic women also frequently exhibits a low lactobacillus distribution, thus resembling that of HPV-infected women, and this likely increases their susceptibility to persistent infections. These findings highlight ethnic variability in microbiota composition and its influence on HPV persistence and cancer progression.
Thus, connecting the fields of microbes and cancers (Microbial Oncogenesis) is a major emerging field with a huge potential for innovation and a target for large-scale funding.
In this context, the University of South Florida (USF) Microbiomes Institute, the USF Virology Institute, and the Tampa General Hospital (TGH) Cancer Institute are launching a consortium which will merge these three highly renowned research and clinical Institutes, it will investigate how the interactions between some cancer-associated viruses and the huge populations of bacteria (named as “microbiomes”) present in the gut and the cancer tissues can favor persistence of chronic viral infections and growth of cancer and its resistance to treatment. The consortium will merge the efforts of the best experts in these different fields of medicine and should offer a transformational approach to cancer treatment and prevention strategies.


Leave a comment